Case Study: Identification of a McLeod phenotype blood donor

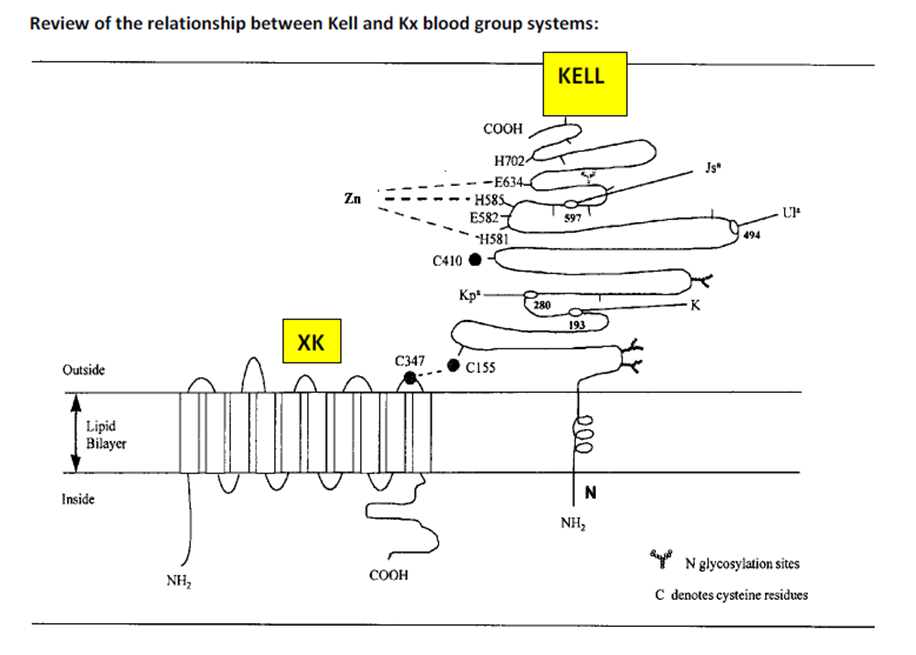
Background/Case Studies: The McLeod phenotype is defined as the absence of the Kx antigen from the red blood cell (RBC) membrane. Kx is the only antigen in the XK blood group system. The Kx antigen is located on the Xk protein, which is linked to the KEL glycoprotein by a single disulfide bond. Absence of the Kx antigen leads to weakened expression of Kell system antigens and acanthocytosis. The McLeod phenotype is X-linked recessive and appears almost exclusively in males. Female carriers of the McLeod phenotype will express mixed RBC populations of normal Kell antigen expression and McLeod phenotype cells. Chronic granulomatous Disease (CGD) has been associated with the McLeod phenotype as they are both induced by genetic deletions on the X chromosome. Those with CGD McLeod type are able to produce anti-KL (anti-Kx + anti-Km). Those of the non-CGD McLeod type are able to produce anti-Km.
Here we will review a case where a McLeod phenotype donor was discovered during routine donor antigen screening in the IRL.
Study Design/Methods: During routine mass antigen screening of donor blood by the American Red Cross Reference Laboratory in Tucson, Arizona, a source of anti-Kpb was included with a batch of units. One donor sample preliminarily tested negative for the Kpb antigen. This donor was additionally tested with a source of anti-Kpa with the expectation the result would be positive, but instead a negative result was obtained. The donor sample was re-tested with anti-K, anti-k, anti-Kpa, anti-Kpb, and anti-Jsb antisera to investigate the irregularity. A negative result was obtained for all testing, except anti-k which produced very weak macroscopic agglutinates. The negative results were examined microscopically for confirmation, at which point microscopic agglutination was noted with anti-Kpb and anti-Jsb. It was also noted during microscopic evaluation that every cell in the field appeared crenated like burr cells. Suspecting sample contamination, a fresh 3% suspension of washed RBCs was prepared, and a drop placed on a slide for microscopic examination. Again, every cell in the fields examined appeared as burr cells.
Due to a lack of rare antisera in the current lab, the unit was shipped to the American Red Cross in Southern California for confirmation of a suspected McLeod phenotype. The remaining donor retention tube was shipped to the National Molecular Lab in Philadelphia for additional investigation.
Results/Findings: Additional testing performed by the Southern California laboratory included antigen typing with a source of anti-k, anti-Kpb, anti-Jsb, and anti-KL. Weakly reactive results were obtained with anti-k, anti-Kpb, and anti-Jsb. A negative result was obtained with anti-KL, serologically confirming the McLeod phenotype. Results of the molecular testing are summarized below.

Conclusions: This donor was confirmed serologically to lack the Kx antigen and have the McLeod phenotype. Multiple known genetic variations can lead to the McLeod phenotype, but this donor did not appear to have any of them. It is possible this donor has a genetic alteration in a region we are currently unable to test for, or it is possible he has a novel genetic mutation leading to the McLeod phenotype. According to medical literature, males with the McLeod phenotype typically have a compensated hemolytic anemia; but this donor had a normal hemoglobin/hematocrit and qualified as a blood donor. Additional follow-up with the donor was performed, along with explanation of his rare type and potential future health complications linked to this particular antigen type. This donor has been able to donate 38 times successfully since the discovery of his rare type in 2012 and further support the American Rare Donor Program. Four of his donations have been used to support a child with McLeod syndrome and CGD during bone marrow transplant.
References:
Reid, Lomas‐Francis. The Blood Group Antigen FactsBook. Third Edition, 2012.
Cohn et al. AABB Technical Manual. Twentieth Edition, 2020.
Russo, DC, et al. First example of anti-Kx in a person with the McLeod phenotype and without chronic granulomatous disease. Transfusion 40, 1371-1375, 2000.
Written By
Karen Rodberg, MBA, MT(ASCP)SBB
Rodberg worked as a Medical Technologist for many years, starting in Minnesota before coming to her senses and moving to California. She began working with a small private hospital in Glendale for 11 years as the Supervisor of the Blood Bank department and performed a wide variety of tasks, from benchwork, to writing procedures and drawing autologous blood donors; but the work that she enjoyed the most was antibody identification. She followed that path and took a job at the American Red Cross in the Immunohematology Reference Lab in 1987, and never looked back. Ms. Rodberg had the privilege to work with Dr. George Garratty for many years, and with Pat Arndt, and Gina Leger. Karen states, “all of them have taught me so much, but the best teacher has been experience. I’ve had the good fortune to see some very interesting cases in our Reference Lab, many due to the reputations of my predecessors.”
Emily Wilson, MT(ASCP)SBB
Wilson is the supervisor of the Immunohematology Reference Laboratories located in Dallas and Houston for the American Red Cross. She began her career with the Red Cross as a medical technologist in the Arizona region, located in Tucson AZ in 2009. In 2014 she relocated to Texas and began working in the laboratory located in Dallas TX. She was promoted to lead technologist, then subsequently to a supervisor position. Currently, she travels to provide oversight for two Texas laboratory sites, continues to perform bench work, provides continuing education and training for internal staff as well as SBB students, and travels to assist with training in other states.